- 11 October 2024
- 1 October 2024
- 1 October 2024
- 30 September 2024
- 20 September 2024
- 20 September 2024
- 22 August 2024
- 8 August 2024
- 2 August 2024
- 29 July 2024
- 16 July 2024
- 19 June 2024
- 8 May 2024
- 17 April 2024
- 5 April 2024
- 4 April 2024
- 28 March 2024
- 27 March 2024
- 21 March 2024
- 1 March 2024
- 1 March 2024
- 1 March 2024
- 5 February 2024
- 23 January 2024
- 16 January 2024
- 21 December 2023
- 17 November 2023
- 15 November 2023
- 1 November 2023
- 25 October 2023
Many biofuels haven’t panned out. Could algae make the clean diesel and aviation fuel Australia needs?
Algal fuels needs much less land than conventional biofuels.
$1.4 million to scale algae biotechnology
Innovative start-up Algenie has won a major national grant to commercialise its biomanufacturing platform.
Healthy eating improves memory, but sugary diets may cause lasting damage
As concern grows about the long‑term health effects of modern diets, new research led by UTS has examined how changes in what we eat affect memory and brain...
Protecting a vulnerable marine icon
A new conservation initiative is bringing together art and science in a bold effort to protect the weedy seadragon, one of Australia’s most extraordinary yet...
E-cigarette devices expose users to toxic metals, UTS study finds
Vaping is largely believed to be a safer alternative to cigarettes, but new research shows that vaping devices can deliver toxic metals directly into lung...
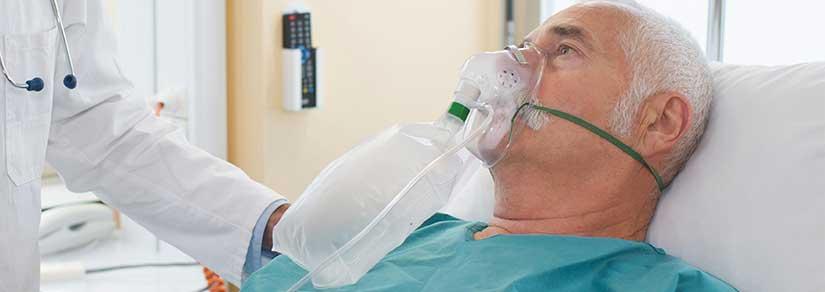
When oil refineries burn, here’s what happens to your lungs and heart
The blaze at the Geelong oil refinery in Victoria has reportedly been extinguished. But residents in the area may be worried about the health effects of...
Major Sydney development a testbed for research on nature positive urban design
Set to become one of Australia's largest urban green roof developments, the Harbourside precinct in Sydney’s Darling Harbour will be the testbed for pioneering...
Need to parent differently now your kid’s a teen or tween? 5 techniques that actually work
Close parent-child relationships reduce the risk of mental health problems and help teenagers cope with stress.
What’s ‘dirty fuel’ doing to our lungs? The same as it did for most of 2025
Australians may have seen headlines warning “dirty fuel” is back, temporarily, to ease fuel supplies. The phrase sounds alarming, but it has a specific and...
Researchers find harmful algae species wasn’t new to South Australian waters
Going back through archived DNA from water samples, scientists have found that the species chiefly responsible for South Australia’s persistent harmful algal...
It’s more than academic for forensics pioneer
Professor Claude Roux recognised with global career-achievement award for his work that has transformed forensic investigation into a true science.
Study reveals the gene networks driving sex differences in respiratory health
A new study has confirmed that male and female lungs are “wired differently” at the molecular level, providing further evidence supporting sex-inclusive...
Award-winning Kamilaroi water scientist charts course for impact
Pioneering hydrogeologist Professor Bradley Moggridge is being acknowledged for his world-leading expertise bridging western and Indigenous science.
Funding boost to help asthma sufferers breathe easier
Dr Chantal Donovan is discovering the unexpected connection between lung and gut health has won $1 million in philanthropic funding.
How the myth of ‘aqua nullius’ still guides Australia’s approach to groundwater
For too long, Indigenous perspectives have not been heard in groundwater science. We must work together to protect Australia’s precious groundwater.
Click go the photons kids, click click click
UTS researchers in the ARC Centre of Excellence for Transformative Meta-Optical Systems (TMOS) are typically working to uncover the applications of light at...
Coral’s harbourside move spawns premier award win
Research tracing the unexpected spread of subtropical coral species in the temperate waters around Sydney is celebrated with a NSW Premier's Prize.
Asbestos has been found in children’s coloured sand. What’s the risk to kids?
Multiple schools and early learning centres in the Australian Capital Territory have shut on Friday after asbestos was found in coloured sand used for...
Species identification key to understanding South Australia’s harmful algal bloom
In a significant step towards dealing with the persistent harmful algal bloom affecting the South Australian coast, researchers have identified the species...
Fish ‘fingerprints’ in the ocean reveal which species are moving homes due to climate change
Species across the planet are on the move. Climate change has already caused more than 12,000 species to shift their homes across land, freshwater and the sea....
3D bioprinted mini placentas could transform pregnancy research
UTS scientists have achieved a major breakthrough by 3D bioprinting miniature placentas, providing a new way to study complications in pregnancy. The research...
Coral research uncovers new species hidden in plain sight
An international team of scientists have made groundbreaking findings that describe five new-to-science species and rewrite the family tree of one of the...
UTS scientists a triple threat for Eurekas
Three UTS researchers have been named as finalists in the 2025 Australian Museum Eureka Prizes, the nation’s highest profile science awards.
Freak wind gusts made worse by climate change threaten airline passenger safety
Unexpected severe turbulence injured crew and passengers on a Qantas Boeing 737 during descent at Brisbane on May 4 2024. The subsequent Australian Transport...
Leading the charge towards a clean energy future
Battery and energy storage pioneer Distinguished Professor Guoxiu Wang has been elected a fellow of the Australian Academy of Science.
Mystery solved: the tiny algae killing fish and harming surfers on SA beaches
Confronting images of dead seadragons, fish and octopuses washed up on South Australian beaches – and disturbing reports of “more than 100” surfers and...
Wild weather linked to weedy seadragon deaths
Marine scientists are calling for beachgoers who find weedy seadragons washed up on the shore to photograph them and send details to researchers at UTS.
Being ready for the worst
Before being confronted with the reality of a disaster, police and emergency services need to be prepared as well as they can be under realistic conditions, an...
The biggest threat at the beach this summer? It’s not sharks
With summer on the way, you might be thinking about your first swim of the season — and perhaps about what lurks beneath the water at your favourite beach.
New research reveals why the Darling River is drying up
Less rain will fall in the Darling River catchment as climate change worsens. This fact must be central to decisions about how much water can be taken from the...
Coral reef destruction a threat to human rights
A human rights-based approach to coral reef protection could ensure governments are held to account for safeguarding marine ecosystems.
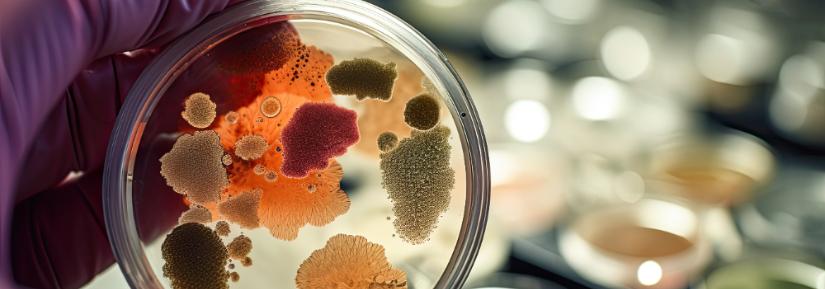
Roundtable Highlights: Addressing Australia’s Biomanufacturing Talent Shortage
The Biologics Innovation Facility (BIF) recently hosted a roundtable with key members of NSW’s biomanufacturing ecosystem, including those from industry,...