Australian researchers who have been working on 3D-printed “mini hearts” to repair damage from cardiovascular disease are refocusing their efforts to help address the immediate threat of COVID-19 to patients with underlying heart disease.
3D-printed ‘mini hearts’ may hold COVID clues
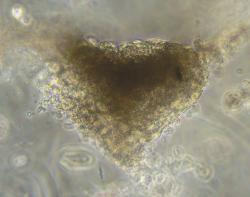
A team at the University of Technology Sydney (UTS) led by bioengineer Dr Carmine Gentile has developed a new technology to generate personalised bioinks made of patient’s own stem cell-derived heart cells as “mini hearts”. These are then used to 3D bioprint cardiac tissues to repair areas of dead tissue in patients with a failing heart.
The scientists now hope these mini hearts will help to understand why COVID-19 patients with heart disease have seven-fold higher mortality compared to other COVID-19 patients. This may provide a platform to test therapies for use in heart disease patients exposed to the potentially deadly coronavirus.
“Together with other colleagues and experts we are seeking to identify what the effects of COVID-19 are on the heart,” Dr Gentile says.
“We have the tools to study how the heart behaves – we can 3D bioprint heart tissues that mimic living heart tissues and use them in a tube to test the effects of drugs and other agents, including viruses.”
Patients who already suffer from cardiovascular disease are known to face a higher chance of dying compared to other patients exposed to the virus.
The current understanding is that the primary site of infection for COVID-19 is the lungs, but the virus can affect other parts of the body, including the heart.
One question is whether COVID-19 is having a direct impact on the heart via the ACE-2 protein, which is considered to be the cellular entry path in the lungs but which is also found in the heart. Alternatively, the virus could be having an indirect impact on the heart due to a systemic inflammatory response.
Our hope is we may be able to identify the mechanisms and help others to develop new therapies
Dr Carmine Gentile
“It is thought that the inflammatory response from the lungs could reach the heart of COVID-19 patients, leading to a deadly effect in the heart,” Dr Gentile says of its impact on cardiovascular disease patients.
“Our hope is we may be able to identify the mechanisms and help others to develop new therapies or even prevent this occurring.”

Dr Carmine Gentile with a 3D bio printer.
Dr Gentile leads the Cardiovascular Regeneration Group at UTS, a multidisciplinary team including experts in cardiovascular biology, bioengineering and biomaterials, along with clinicians such as cardiologists and cardiothoracic surgeons, who are seeking to better deliver personalised therapies for patients suffering heart failure.
Their 3D bioprinted, patient-specific cardiac patches are now in pre-clinical trials – required before they can be tested in humans – and are currently being used in the lab for drug testing to prevent side effects of drugs on patients’ hearts.
Cardiovascular disease is one of the leading causes of death worldwide. In Australia, it accounts for about one in four deaths – claiming a life every 13 minutes.
Heart failure is a frequent complication of heart disease, with inadequate blood supply resulting in the death of heart tissue in an affected area. Life-long drug therapy may be required as a result, and end-stage heart failure may mean a patient joins the waiting list for a heart transplant.
Stem cell therapy has been a recent area of investigation for the treatment of heart failure but faces hurdles such as a body rejecting cells from a donor.
Dr Gentile’s approach tries to replicate the natural development of heart tissue, as observed in embryos and babies, to create a complex but “friendlier” environment for cells that are sourced from the patient themselves.
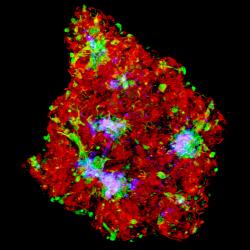
This pathway to fully functional heart tissue involves bespoke “bioink” being 3D printed onto specialist “biopaper”, to form cardiac patches specific to each patient.
“The materials we are growing cells in, in bioprinted heart tissues, should match the features typical of the real heart,” Dr Gentile says.
In the first stage of the process, blood or skin cells are collected from the patient and reprogrammed to make stem cells. “From these stem cells we make the heart cells,” he explains. “With these heart cells we become the ‘Masterchefs’ of cardiovascular disease, mixing them together with other biocompatible components to create the bioink.”
Meanwhile, a computerised tomography (CT) scan provides a 3D image of the individual patient’s heart, and magnetic resonance imaging (MRI) reveals the details of the damage that needs patching.
At the 3D bioprinter, the information from the CT scan and MRI guides layer-on-layer deposits of the bioink onto the biopaper – a biocompatible gel, or hydrogel, that has features such as high water content to allow the cells to live and function properly.
The result is a three-dimensional patch of heart tissue specific to the patient and to the area of damage.
Dr Gentile hopes that, one day, the surgeon themselves may be able to independently 3D bioprint this tissue in the operating theatre.
For now, the next step is to quickly move to clinical trials of the cardiac patches. “Our main goals are to make sure these bioprinted heart tissues are safe and can improve the condition of a failing heart,” Dr Gentile says.
In the lab, mini hearts produced via the same process are already being used to test drug therapies. The team has used the technology to examine the toxic effects of doxorubicin, a drug that is used to treat breast cancer in women and leukaemia and lymphoma in children.
“The drug works to kill the cancer but it has a very high toxicity for the heart,” Dr Gentile explains. “We’ve been able to establish how this has been happening using the bioprinted mini hearts. Now we want to experiment how COVID-19 affects the heart, by using bioprinted mini hearts using a similar approach.”
So, an innovation aimed at preventing the death of many Australians from cardiovascular disease may well help save lives from a new and very different threat.
Read a research paper on this topic

Long interview with Carmine Gentile
